5.02 Quiz Balancing Chemical Equations
11.3: Balancing Equations
- Page ID
- 53781

Any leftovers?
When yous cook a repast, quite often in that location are leftovers because you lot prepared more than people would eat at one sitting. Sometimes when y'all repair a piece of equipment, you end up with what are chosen "pocket parts"—small pieces you put in your pocket considering y'all're non certain where they belong. Chemical science tries to avert leftovers and pocket parts. In normal chemical processes, we cannot create or destroy matter (police force of conservation of mass). If we start out with ten carbon atoms, we need to end up with ten carbon atoms. John Dalton's atomic theory said that chemical reactions basically involve the rearrangement of atoms. Chemical equations need to follow these principles in guild to be right.
Balancing Chemic Equations
A counterbalanced equation is a chemical equation in which mass is conserved and at that place are equal numbers of atoms of each element on both sides of the equation. We tin write a chemical equation for the reaction of carbon with hydrogen gas to form methane \(\left( \ce{CH_4} \right)\):
\[\brainstorm{array}{ccccc} \ce{C} \left( s \right) & + & \ce{H_2} \left( g \right) & \rightarrow & \ce{CH_4} \left( g \right) \\ 2 \: \ce{C} \: \text{atoms} & & 2 \: \ce{H} \: \text{atoms} & & ane \: \ce{C} \: \text{cantlet,} \: iv \: \ce{H} \: \text{atoms} \terminate{array}\nonumber \]
In order to write a right equation, you must offset write the right skeleton equation with the right chemical formulas. Retrieve that hydrogen is a diatomic molecule and and then is written as \(\ce{H_2}\).
When we count the number of atoms of both elements, shown under the equation, we see that the equation is not counterbalanced. There are only 2 atoms of hydrogen on the reactant side of the equation, while in that location are 4 atoms of hydrogen on the product side. We can balance the above equation by adding a coefficient of ii in front of the formula for hydrogen.
\[\ce{C} \left( s \right) + 2 \ce{H_2} \left( g \right) \rightarrow \ce{CH_4} \left( yard \right)\nonumber \]
A coefficient is a modest whole number placed in front of a formula in an equation in social club to residuum it. The 2 in forepart of the \(\ce{H_2}\) means that there are a total of \(2 \times 2 = 4\) atoms of hydrogen as reactants. Visually, the reaction looks like the figure below.
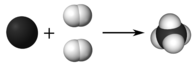
In the balanced equation, there is one atom of carbon and iv atoms of hydrogen on both sides of the arrow. Below are guidelines for writing and balancing chemical equations.
- Make up one's mind the right chemical formulas for each reactant and production.
- Write the skeleton equation.
- Count the number of atoms of each element that appears equally a reactant and as a product. If a polyatomic ion is unchanged on both sides of the equation, count it equally a unit.
- Balance each element ane at a fourth dimension by placing coefficients in front of the formulas.
- It is best to begin by balancing elements that simply appear in one chemic formula on each side of the equation.
- No coefficient is written for a 1.
- NEVER change the subscripts in a chemic formula—you tin can just balance equations past using coefficients.
- Bank check each atom or polyatomic ion to be certain that they are equal on both sides of the equation.
- Make sure that all coefficients are in the lowest possible ratio. If necessary, reduce to the everyman ratio.
Case \(\PageIndex{ane}\): Balancing Chemic Equations
Aqueous solutions of atomic number 82 (II) nitrate and sodium chloride are mixed. The products of the reaction are an aqueous solution of sodium nitrate and a solid precipitate of pb (II) chloride. Write the balanced chemical equation for this reaction.
Solution
Footstep i: Plan the problem.
Follow the steps for writing and balancing a chemical equation listed in the text.
Step 2: Solve.
Write the skeleton equation with the right formulas.
\[\ce{Lead(NO_3)_2} \left( aq \right) + \ce{NaCl} \left( aq \right) \rightarrow \ce{NaNO_3} \left( aq \correct) + \ce{PbCl_2} \left( south \right)\nonumber \]
Count the number of each atom or polyatomic ion on both sides of the equation.
\[\brainstorm{assortment}{ll} \textbf{Reactants} & \textbf{Products} \\ one \: \ce{Pb} \: \text{atom} & i \: \ce{Pb} \: \text{cantlet} \\ 2 \: \ce{NO_3^-} \: \text{ions} & one \: \ce{NO_3^-} \: \text{ions} \\ one \: \ce{Na} \: \text{atom} & 1 \: \ce{Na} \: \text{atom} \\ 1 \: \ce{Cl} \: \text{cantlet} & two \: \ce{Cl} \: \text{atoms} \terminate{array}\nonumber \]
The nitrate ions and the chlorine atoms are unbalanced. Start past placing a two in front of the \(\ce{NaCl}\). This increases the reactant counts to 2 \(\ce{Na}\) atoms and 2 \(\ce{Cl}\) atoms. And then place a 2 in front of the \(\ce{NaNO_3}\). The result is:
\[\ce{Pb(NO_3)_2} \left( aq \correct) + 2 \ce{NaCl} \left( aq \right) \rightarrow 2 \ce{NaNO_3} \left( aq \right) + \ce{PbCl_2} \left( s \right)\nonumber \]
The new count for each atom and polyatomic ion becomes:
\[\brainstorm{assortment}{ll} \textbf{Reactants} & \textbf{Products} \\ 1 \: \ce{Pb} \: \text{cantlet} & 1 \: \ce{Pb} \: \text{atom} \\ two \: \ce{NO_3^-} \: \text{ions} & two \: \ce{NO_3^-} \: \text{ions} \\ ii \: \ce{Na} \: \text{atom} & two \: \ce{Na} \: \text{atom} \\ 2 \: \ce{Cl} \: \text{cantlet} & 2 \: \ce{Cl} \: \text{atoms} \end{array}\nonumber \]
Step 3: Think about your outcome.
The equation is now counterbalanced since in that location are equal numbers of atoms of each element on both sides of the equation.
Summary
- The procedure of balancing chemical equations is described.
Review
- What is the police force of conservation of mass?
- How did Dalton describe the procedure of a chemical reaction?
- Why don't we change the subscripts in order to balance an equation?
5.02 Quiz Balancing Chemical Equations,
Source: https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Introductory_Chemistry_(CK-12)/11%3A_Chemical_Reactions/11.03%3A_Balancing_Equations
Posted by: davisshmed1990.blogspot.com


0 Response to "5.02 Quiz Balancing Chemical Equations"
Post a Comment